Throughout recent years, the Kerr Group Research Team have developed a portfolio of research activities that have led to contributions to knowledge and the development of new methods in a number of individual preparative fields and, in particular, within the broad area of metal-mediated processes as applied to organic synthesis and asymmetric transformations. The emerging techniques from the Kerr Group Research laboratories have also been applied in a range of total synthesis programmes.
Key current topics are described below:
Organoiridium Catalysis
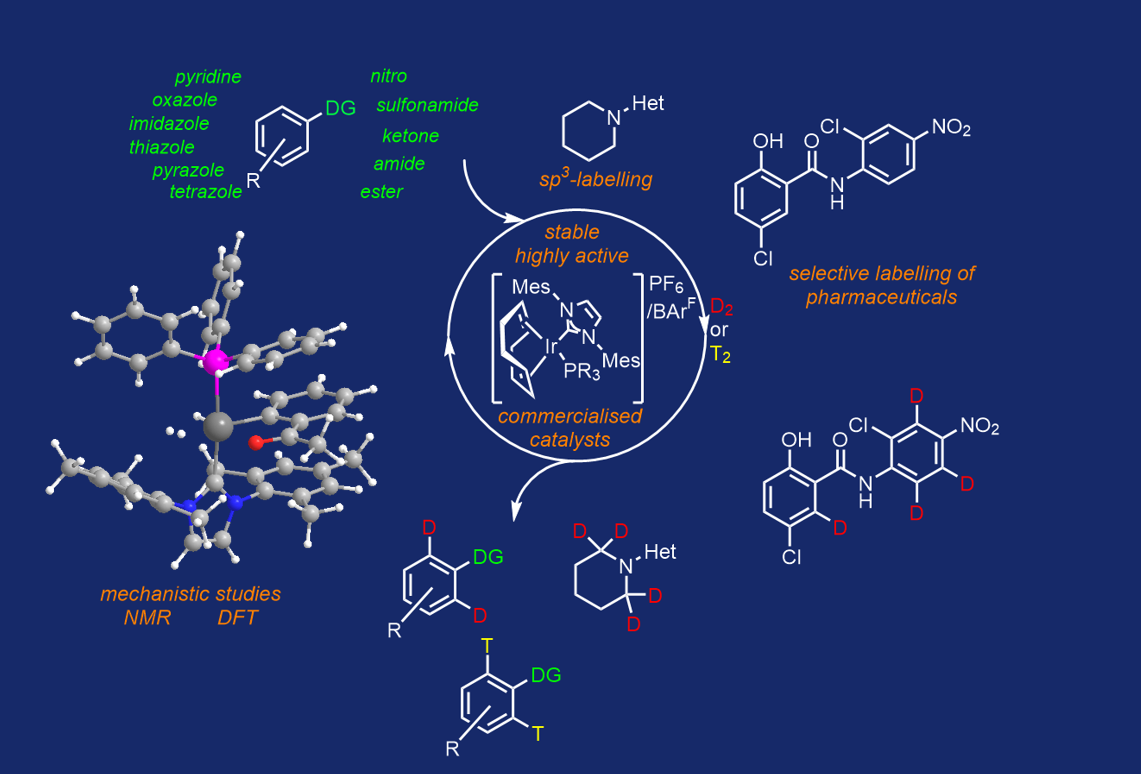
A suite of new iridium C-H activation catalysts has been designed and prepared and which now display leading levels of reactivity and selectivity as related to hydrogen-isotope exchange reactions with a wide variety of organic substrates. This means that valuable iridium complexes can now be used at significantly lower loading and under notably mild reaction conditions. These exchange reactions are of great importance to pharmaceutical industries, where labelled versions of pharmaceutical candidate compound and their metabolites are in constant demand, in order to accelerate the understanding of the metabolism and other properties of potential drug species.
The Kerr Group Research team are now expanding their array of new catalysts developed within their laboratories, and attempting to deepen their mechanistic understanding of the iridium-catalysed hydrogen-isotope exchange reactions, as well as extending the application of the emerging complexes across an additional portfolio of important transformations in organic synthesis, such as hydrogenation, and C-C, C-N and C-O bond forming processes.
Computational Chemistry
It is important to note that our group’s success in the organoiridium catalysis area has been appreciably facilitated by the synergistic use of computational chemistry to inform reaction mechanism and catalyst design, whilst driving the expansion of the reaction scope. Our researchers blend theoretical studies with preparative endeavours across all projects.
Commercialised Catalysts
A number of our iridium species as applied in catalysis processes developed in our laboratory have been commercialised by Strem Chemicals, Inc.
Organocobalt Chemistry – Development and Applications of the Pauson-Khand Reaction
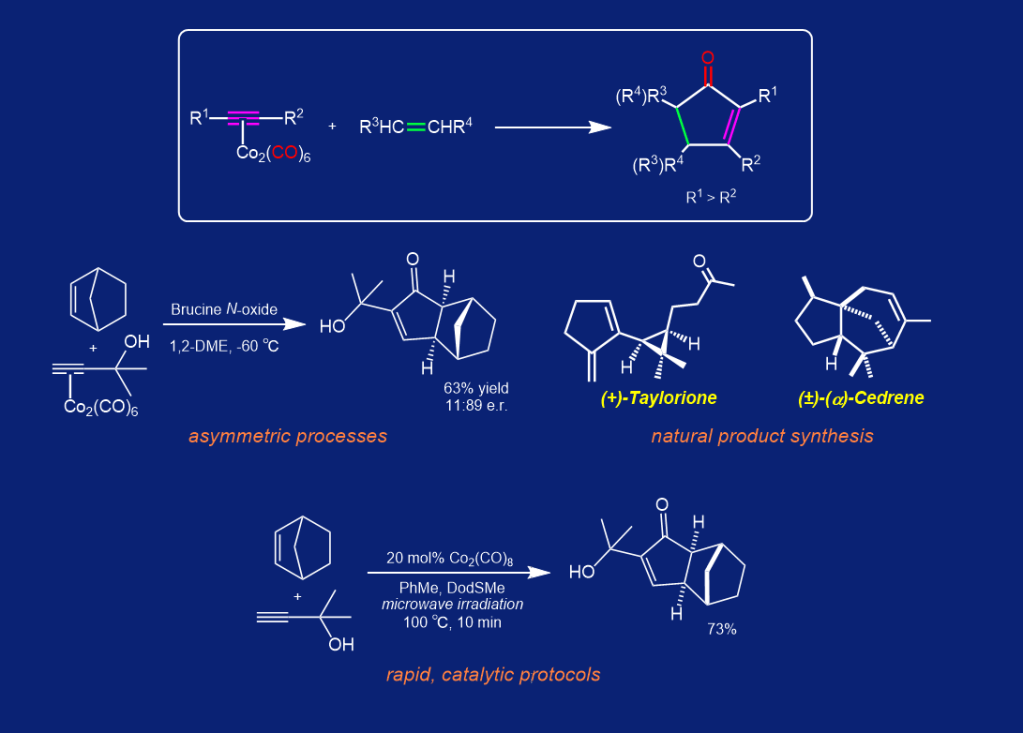
Research within our laboratories has been geared towards furthering the scope and synthetic utility of the Pauson-Khand (P-K) reaction. In addition to applying the reaction within the syntheses of a spectrum of natural products, such as Taylorione and Cedrene, we have contributed a number of additional developments within the P-K area, including studies on the first direct asymmetric P-K reaction, advances in solid supported promotion conditions, use of non-gaseous ethylene equivalents, investigations into controlling the regiochemical outcome of the olefin insertion step, development of efficient ultrasound promotion methods, and, more recently, a completely odourless and more economical sulfide-promoted protocol.
Current research aims to further develop the effectiveness of the P-K reaction towards delivering a wider range of desirable and elaborated chemical scaffolds. More specifically, we are probing alternative functionalised alkene components to the more standard and typically employed olefin substrates.
Organomagnesium Chemistry

The production of enantiomerically-enriched products is of continued global importance; this is key to the manufacture of safe and marketable materials and also leads to less waste in terms of energy, solvents, and unwanted (enantiomeric) by-products.
In this field of research, we have developed a range of chiral magnesium amide bases for the asymmetric deprotonation of prochiral ketones. Initially, we established an effective process for the formation of chiral silyl enol ethers and demonstrated that such protocols are intrinsically simpler and more generally effective with organomagnesium-based methods than with the earlier developed lithium-based systems. The optimised levels of asymmetric induction are often enhanced over that achieved with the more practically demanding lithium chemistry, and the enantioselective processes can be performed at more accessible temperatures (compared to the typically cryogenic conditions required for their organolithium counterparts).
Further to this, our team have extended this methodology to the preparation of optically active enol phosphates. We are currently investigating the application of this chemistry towards the preparation of novel bicyclic, sp3-rich scaffolds, which are desirable synthetic targets within the pharmaceutical industry due to their favourable physical properties and occupation of underexplored regions of chemical space.
Pharmaceutical partner-aligned Research Programmes
The Kerr Group Research team has strong links with industrial partners. Indeed, approximately two-thirds of Prof. Kerr’s supervised PhD students have been in collaboration with industry, which exemplifies the relevance of the research team’s endeavours to both industrial and academic communities.
In addition to the synthetic organic chemistry and catalysis aspects of our group’s expertise, Prof. Kerr has been the academic lead on an array of medicinally-aligned research collaborations, with a track record of success in these areas. On-going projects in collaboration with pharmaceutical industry partners, include:
- new reagents for late-stage aromatic labelled methylation methods (GSK);
- sulfonylative cross-coupling reactions for the preparation of medicinally relevant small molecules (GSK);
- formation of pharmaceutically relevant functionality through new electrochemically-driven methods (GSK);
- enantioselective trifluoromethylation reactions (GSK);
- application of alkyl trifluoroboronates to sp3-sp3 couplings and expansion to enantioenriched substrates within palladium-catalysed methods as applied to drug discovery programmes (GSK); and
- protein degradation: PROTAC technology for central nervous system penetration and inhaled delivery (Charles River Laboratories)
Funding
The research work within the WJK research group has been supported, and continues to be supported, by the EPSRC, private funding bodies (such as the Carnegie Trust, Medical Research Scotland and the C. K. Marr Trust), and a range of industrial companies. Past and present industrial collaborators include: GlaxoWellcome/GlaxoSmithKline, AstraZeneca, Pfizer, MerckSharp&Dohme, Sanofi-Synthelabo, Lancaster Synthesis Ltd., Celltech-Chiroscience, and Organon/Schering-Plough.





